Abstract
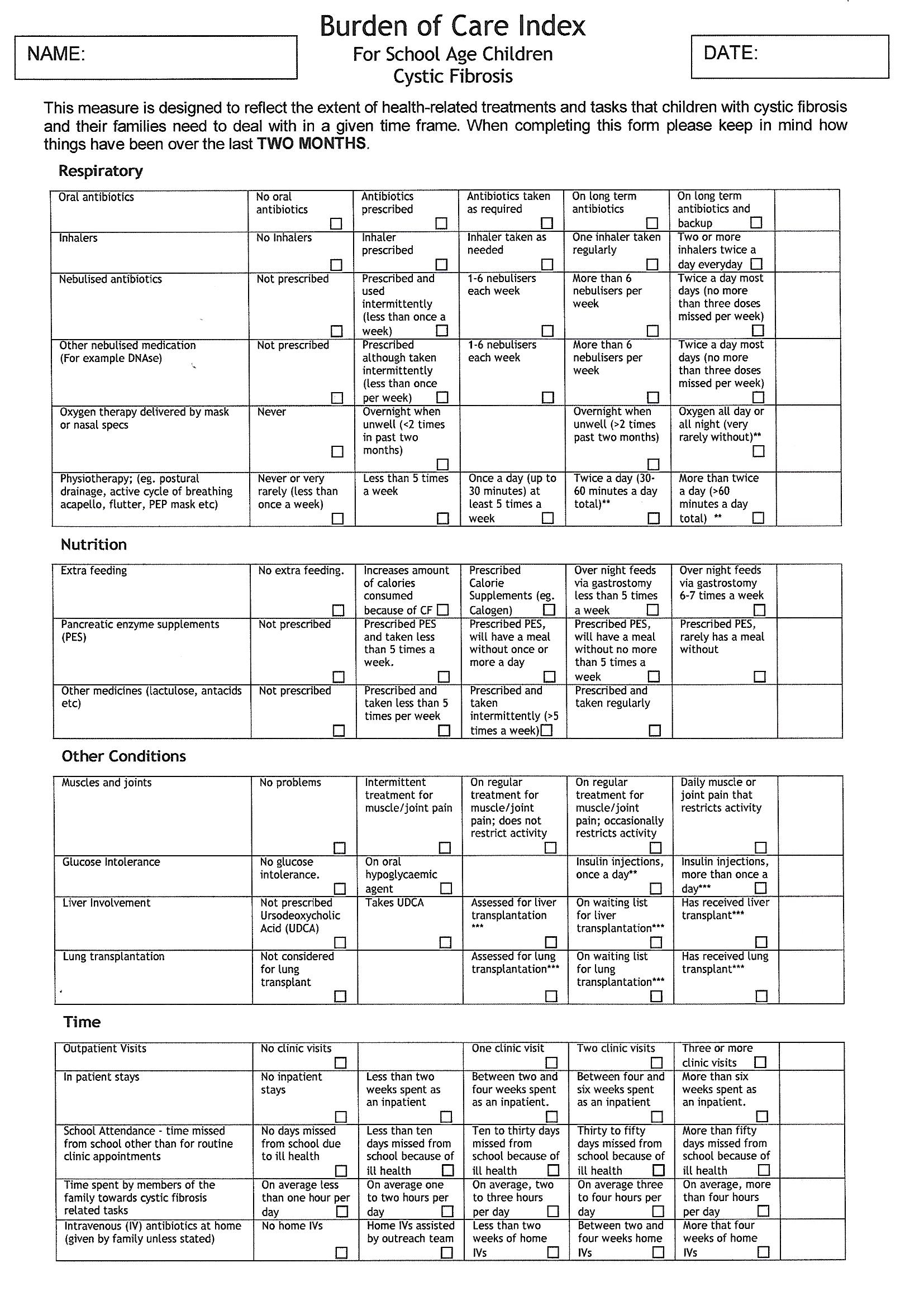
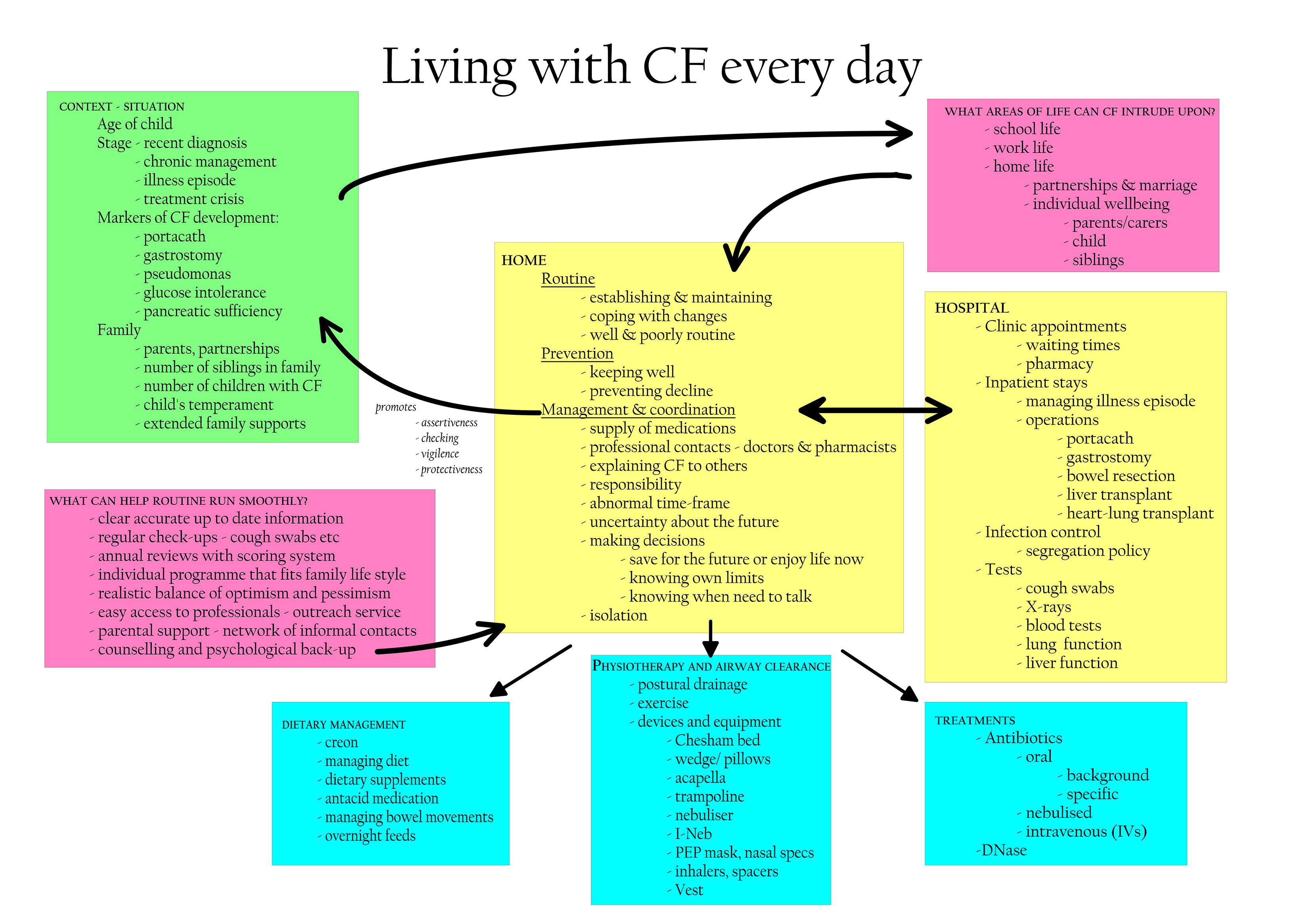
OBJECTIVE: Treatments for cystic fibrosis (CF) are complex, labour-intensive, and perceived as highly burdensome by caregivers of children with CF. An instrument assessing burden of care is needed. DESIGN: A stepwise, qualitative design was used to create the CLCF with caregiver focus groups, participant researchers, a multidisciplinary professional panel, and cognitive interviews. MAIN OUTCOME MEASURES: Preliminary psychometric analyses evaluated the reliability and convergent validity of the CLCF scores. Cronbach's alpha assessed internal consistency and t-tests examined test-retest reliability. Correlations measured convergence between the Treatment Burden scale of the Cystic Fibrosis Questionnaire-Revised (CFQ-R) and the CLCF. Discriminant validity was assessed by comparing CLCF scores in one vs two-parent families, across ages, and in children with vs without Pseudomonas aeruginosa (PA). RESULTS: Six Challenge subscales emerged from the qualitative data and the professional panel constructed a scoresheet estimating the Time and Effort required for treatments. Internal consistency and test-retest reliability were adequate. Good convergence was found between the Total Challenge score and Treatment Burden on the CFQ-R (r=-0.49, p?=?0.02, n?=?31). A recent PA infection signalled higher Total Challenge for caregivers (F(23)11.72, p?=?0.002). CONCLUSIONS: The CLCF, developed in partnership with parents/caregivers and CF professionals, is a timely, disease-specific burden measure for clinical research.